Research
Structure-function relationships of CO2 reduction electrocatalysts at work
The electrocatalytic reduction of CO2 into hydrocarbon fuels, like methane or ethylene, is regarded as a promising strategy to address one of the main current environmental issues: reducing the CO2 footprint of our society. However, improvements in activity, selectivity and stability of the developed electrocatalysts are crucial in order to implement electrocatalysis on a large scale. Colloidal synthesis of metal nanoparticles offers a versatile strategy to boost the activity (large surface area) and selectivity (selective facet exposure) of the CO2 reduction reaction, but is limited by nanoparticle destabilization under reaction conditions. Therefore, detailed characterization over multiple length- and timescales is required to elucidate the reaction mechanism of CO2 reduction electrocatalysts at work
Our research focuses on (1) the synthesis of colloidal metal electrocatalyst nanoparticles with well-defined sizes, shapes and compositions (e.g. nanorods or nanoplatelets), (2) in situ vibrational spectroscopy to study the adsorbed intermediates at the catalyst surface in space and time, and (3) in situ X-ray characterization (diffraction, spectroscopy, scattering) to elucidate the structure of the electrocatalyst under working conditions (Figure 1). Our main interest lies in unravelling structure-function relationships by performing in situ X-ray diffraction and scattering experiments, as well as in situspectroscopy measurements. Colloidal nanomaterials are ideally suited for structure-function relationships, since they can be prepared with atomic precision in solution. This not only allows us to deposit them on various electrodes, but also characterize the size, shape and faceting during the reaction and use these parameters to direct the formation of value-added chemicals, such as C>2 hydrocarbons. These in situ characterization techniques will give valuable fundamental, but also practical insights into the exact reaction mechanism of the CO2 reduction reaction and the (de)activation of the electrocatalyst nanoparticles, which will allow us to rationally design the ultimate electrocatalyst.
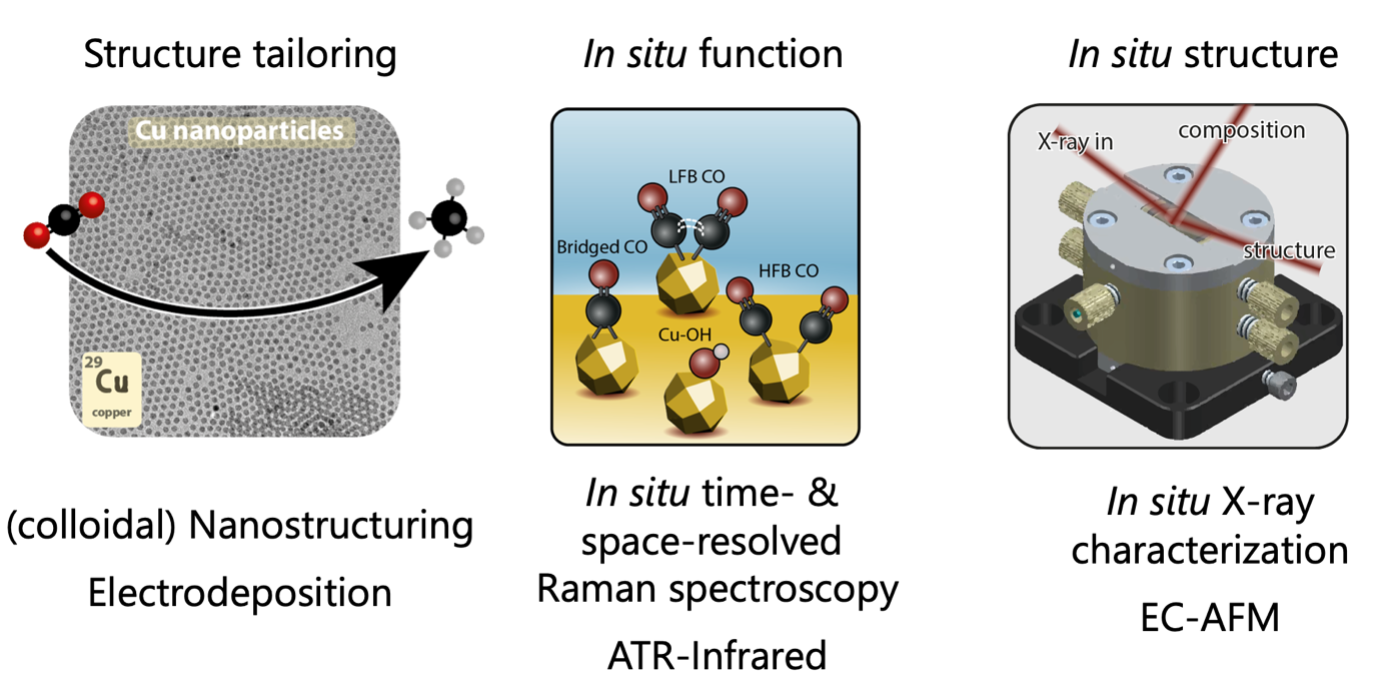
Figure 1. (left) Colloidal nanostructures ooffer a versatile strategy to steer the CO2 reduction reaction. (middle) in situ vibrational spectroscopy (Raman and Infrared) sheds light on adsorbed reaction intermediates at the catalyst surface. (right) in situ X-ray characterization is utilized to unravel the structure (size, shape, faceting, composition) of the colloidal electrocatalysts under working conditions and elucidate (de)stabilization parameters. The obtained information from these three pillars will be used to design the ultimate active, selective and stable nanoparticle electrocatalyst
CV
Tenure Track Assistant Professor (June 2019 – Present), Utrecht University, The Netherlands
Postdoctoral Researcher (March 2019 – June 2019), Utrecht University, The Netherlands
Research topics: Electrocatalytic reduction of CO2 with colloidal metal nanoparticles, under the supervision of prof. dr. ir. Bert M. Weckhuysen
Postdoctoral Researcher (October 2016 – February 2019), Delft University of Technology, The Netherlands
Research topics: Electrochemical doping of semiconductor nanocrystals, (spectro)electrochemistry, ultrafast
spectroscopy, in-situ EXAFS, synthesis of colloidal perovskite nanoplatelets, streak camera measurements, charge transfer and excited state dynamics, transient absorption spectroscopy, under the supervision of dr. Arjan Houtepen and dr. Ferdinand Grozema
PhD student (October 2012 – September 2016), Utrecht University, The Netherlands
Thesis entitled: Tailoring on the nanoscale: control over size, shape, composition and self-assembly of copper chalcogenide nanocrystals
Research topics: Nanocrystal synthesis, cation exchange, self-assembled superstructures, optical spectroscopy, in-situ X-ray scattering, Electron Microscopy, under the supervision of dr. Celso de Mello Donegá and prof. dr. Andries Meijerink
MSc Chemistry & Physics (September 2010 – October 2012), Utrecht University, The Netherlands
Thesis entitled: Colloidal Nanostructures for Application in Quantum Dot Sensitized Solar Cells, under the supervision of dr. Celso de Mello Donegá
MSc Internship(March 2012 – September 2012), Istituto Italiano di Tecnologia, Genova, Italy
Internship report entitled: Control over different colloidal nanocrystal syntheses; size, shape and properties, under the supervision of prof. dr. Liberato Manna
BSc Chemistry (September 2007 – September 2010), Utrecht University, The Netherlands
Thesis entitled: Quantum Dot Sensitized Solar Cells, under the supervision of dr. Esther Groeneveld and dr. Celso de Mello Donegá
Born in Nieuwegein, The Netherlands 1989
Wang, Hui; Mandemaker, Laurens D. B.; Ruiter, Jim; Yu, Xiang; Stam, Ward; Weckhuysen, Bert M.
Identical Grain Atomic Force Microscopy Elucidates Facet-Dependent Restructuring of Copper for CO2 Electroreduction Journal Article
In: Angewandte Chemie - International Edition, vol. 64, no. 16, 2025, (Cited by: 0; All Open Access, Hybrid Gold Open Access).
@article{Wang2025,
title = {Identical Grain Atomic Force Microscopy Elucidates Facet-Dependent Restructuring of Copper for CO2 Electroreduction},
author = {Hui Wang and Laurens D. B. Mandemaker and Jim Ruiter and Xiang Yu and Ward Stam and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-105002639129&doi=10.1002%2fanie.202424530&partnerID=40&md5=331c22c2fbf34965b1ee5ce8b834b351},
doi = {10.1002/anie.202424530},
year = {2025},
date = {2025-01-01},
journal = {Angewandte Chemie - International Edition},
volume = {64},
number = {16},
note = {Cited by: 0; All Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Srivastava, Ketki; Jacobs, Thimo S.; Ostendorp, Stefan; Jonker, Dirk; Brzesowsky, Floor A.; Susarrey-Arce, Arturo; Gardeniers, Han; Wilde, Gerhard; Weckhuysen, Bert M.; Berg, Albert; Stam, Ward; Odijk, Mathieu
Alternative nano-lithographic tools for shell-isolated nanoparticle enhanced Raman spectroscopy substrates Journal Article
In: Nanoscale, vol. 23, 2024, (Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Srivastava2024,
title = {Alternative nano-lithographic tools for shell-isolated nanoparticle enhanced Raman spectroscopy substrates},
author = {Ketki Srivastava and Thimo S. Jacobs and Stefan Ostendorp and Dirk Jonker and Floor A. Brzesowsky and Arturo Susarrey-Arce and Han Gardeniers and Gerhard Wilde and Bert M. Weckhuysen and Albert Berg and Ward Stam and Mathieu Odijk},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85188521560&doi=10.1039%2fd4nr00428k&partnerID=40&md5=06132ad0d716d4e2bf07d5202ec915e2},
doi = {10.1039/d4nr00428k},
year = {2024},
date = {2024-01-01},
journal = {Nanoscale},
volume = {23},
note = {Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Jacobs, Thimo S.; Park, Sunghak; Schönig, Marco; Weckhuysen, Bert M.; Koper, Marc T. M.; Stam, Ward
Luminescence Thermometry Probes Local Heat Effects at the Platinum Electrode Surface during Alkaline Water Electrolysis Journal Article
In: ACS Energy Letters, vol. 9, no. 7, pp. 3335 – 3341, 2024, (Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Jacobs20243335,
title = {Luminescence Thermometry Probes Local Heat Effects at the Platinum Electrode Surface during Alkaline Water Electrolysis},
author = {Thimo S. Jacobs and Sunghak Park and Marco Schönig and Bert M. Weckhuysen and Marc T. M. Koper and Ward Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85196420857&doi=10.1021%2facsenergylett.4c01238&partnerID=40&md5=cd3ccbbc21061e2aa390e20681434150},
doi = {10.1021/acsenergylett.4c01238},
year = {2024},
date = {2024-01-01},
journal = {ACS Energy Letters},
volume = {9},
number = {7},
pages = {3335 – 3341},
note = {Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Kok, Jesse; Ruiter, Jim; Stam, Ward; Burdyny, Thomas
Interrogation of Oxidative Pulsed Methods for the Stabilization of Copper Electrodes for CO2 Electrolysis Journal Article
In: Journal of the American Chemical Society, 2024, (Cited by: 4; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Kok2024,
title = {Interrogation of Oxidative Pulsed Methods for the Stabilization of Copper Electrodes for CO2 Electrolysis},
author = {Jesse Kok and Jim Ruiter and Ward Stam and Thomas Burdyny},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85198172889&doi=10.1021%2fjacs.4c06284&partnerID=40&md5=673919980adb45f3601a5232f4c298d7},
doi = {10.1021/jacs.4c06284},
year = {2024},
date = {2024-01-01},
journal = {Journal of the American Chemical Society},
note = {Cited by: 4; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Srivastava, K.; Boyle, N. D.; Flaman, G. T.; Ramaswami, B.; van den Berg, A.; van der Stam, W.; Burgess, I. J.; Odijk, M.
In situ spatiotemporal characterization and analysis of chemical reactions using an ATR-integrated microfluidic reactor Journal Article
In: Lab on a Chip, vol. 23, no. 21, pp. 4690 – 4700, 2023, (Cited by: 0; All Open Access, Hybrid Gold Open Access).
@article{Srivastava20234690,
title = {In situ spatiotemporal characterization and analysis of chemical reactions using an ATR-integrated microfluidic reactor},
author = { K. Srivastava and N.D. Boyle and G.T. Flaman and B. Ramaswami and A. van den Berg and W. van der Stam and I.J. Burgess and M. Odijk},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85174524108&doi=10.1039%2fd3lc00521f&partnerID=40&md5=275477db76b50cda392194fce4ccc9c6},
doi = {10.1039/d3lc00521f},
year = {2023},
date = {2023-01-01},
journal = {Lab on a Chip},
volume = {23},
number = {21},
pages = {4690 – 4700},
note = {Cited by: 0; All Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Jacobs, Thimo S.; van Swieten, Thomas P.; Vonk, Sander J. W.; Bosman, Isa P.; Melcherts, Angela E. M.; Janssen, Bas C.; Janssens, Joris C. L.; Monai, Matteo; Meijerink, Andries; Rabouw, Freddy T.; van der Stam, Ward; Weckhuysen, Bert M.
Mapping Temperature Heterogeneities during Catalytic CO2 Methanation with Operando Luminescence Thermometry Journal Article
In: ACS Nano, vol. 17, no. 20, pp. 20053 – 20061, 2023, (Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Jacobs202320053,
title = {Mapping Temperature Heterogeneities during Catalytic CO2 Methanation with Operando Luminescence Thermometry},
author = { Thimo S. Jacobs and Thomas P. van Swieten and Sander J. W. Vonk and Isa P. Bosman and Angela E. M. Melcherts and Bas C. Janssen and Joris C. L. Janssens and Matteo Monai and Andries Meijerink and Freddy T. Rabouw and Ward van der Stam and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85175270615&doi=10.1021%2facsnano.3c05622&partnerID=40&md5=79e89f318447b6d0adbcc7f35ef6bb20},
doi = {10.1021/acsnano.3c05622},
year = {2023},
date = {2023-01-01},
journal = {ACS Nano},
volume = {17},
number = {20},
pages = {20053 – 20061},
note = {Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Jonker, Dirk; Srivastava, Ketki; Lafuente, Marta; Susarrey-Arce, Arturo; van der Stam, Ward; van den Berg, Albert; Odijk, Mathieu; Gardeniers, Han J. G. E
Low-Variance Surface-Enhanced Raman Spectroscopy Using Confined Gold Nanoparticles over Silicon Nanocones Journal Article
In: ACS Applied Nano Materials, vol. 6, no. 11, pp. 9657 – 9669, 2023, (Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Jonker20239657,
title = {Low-Variance Surface-Enhanced Raman Spectroscopy Using Confined Gold Nanoparticles over Silicon Nanocones},
author = { Dirk Jonker and Ketki Srivastava and Marta Lafuente and Arturo Susarrey-Arce and Ward van der Stam and Albert van den Berg and Mathieu Odijk and Han J.G.E Gardeniers},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85162927251&doi=10.1021%2facsanm.3c01249&partnerID=40&md5=ca47888d7113fe04de9ed16838f59dff},
doi = {10.1021/acsanm.3c01249},
year = {2023},
date = {2023-01-01},
journal = {ACS Applied Nano Materials},
volume = {6},
number = {11},
pages = {9657 – 9669},
note = {Cited by: 0; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Vos, Rafaël E.; Kolmeijer, Kees E.; Jacobs, Thimo S.; Stam, Ward Van Der; Weckhuysen, Bert M.; Koper, Marc T. M.
How Temperature Affects the Selectivity of the Electrochemical CO2Reduction on Copper Journal Article
In: ACS Catalysis, vol. 13, no. 12, pp. 8080 – 8091, 2023, (Cited by: 12; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Vos20238080,
title = {How Temperature Affects the Selectivity of the Electrochemical CO2Reduction on Copper},
author = { Rafaël E. Vos and Kees E. Kolmeijer and Thimo S. Jacobs and Ward Van Der Stam and Bert M. Weckhuysen and Marc T. M. Koper},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85162895552&doi=10.1021%2facscatal.3c00706&partnerID=40&md5=f9dacee7076d71ad4fbf8bc7d7f386e8},
doi = {10.1021/acscatal.3c00706},
year = {2023},
date = {2023-01-01},
journal = {ACS Catalysis},
volume = {13},
number = {12},
pages = {8080 – 8091},
note = {Cited by: 12; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Yang, Shuang; An, Hongyu; Arnouts, Sven; Wang, Hui; Yu, Xiang; de Ruiter, Jim; Bals, Sara; Altantzis, Thomas; Weckhuysen, Bert M.; van der Stam, Ward
Halide-guided active site exposure in bismuth electrocatalysts for selective CO2 conversion into formic acid Journal Article
In: Nature Catalysis, vol. 6, no. 9, pp. 796 – 806, 2023, (Cited by: 9; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Yang2023796,
title = {Halide-guided active site exposure in bismuth electrocatalysts for selective CO2 conversion into formic acid},
author = { Shuang Yang and Hongyu An and Sven Arnouts and Hui Wang and Xiang Yu and Jim de Ruiter and Sara Bals and Thomas Altantzis and Bert M. Weckhuysen and Ward van der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85168155280&doi=10.1038%2fs41929-023-01008-0&partnerID=40&md5=f872df97069f25c7535feb292dda90c0},
doi = {10.1038/s41929-023-01008-0},
year = {2023},
date = {2023-01-01},
journal = {Nature Catalysis},
volume = {6},
number = {9},
pages = {796 – 806},
note = {Cited by: 9; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
den Hollander, Jan; van der Stam, Ward
In situ spectroscopy and diffraction to look inside the next generation of gas diffusion and zero-gap electrolyzers Journal Article
In: Current Opinion in Chemical Engineering, vol. 42, 2023, (Cited by: 0).
@article{denHollander2023,
title = {In situ spectroscopy and diffraction to look inside the next generation of gas diffusion and zero-gap electrolyzers},
author = { Jan den Hollander and Ward van der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85175469968&doi=10.1016%2fj.coche.2023.100979&partnerID=40&md5=c03ed37b747d84dc8658232e40ff5a5b},
doi = {10.1016/j.coche.2023.100979},
year = {2023},
date = {2023-01-01},
journal = {Current Opinion in Chemical Engineering},
volume = {42},
note = {Cited by: 0},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Monai, Matteo; Jenkinson, Kellie; Melcherts, Angela E. M.; Louwen, Jaap N.; Irmak, Ece A.; Aert, Sandra Van; Altantzis, Thomas; Vogt, Charlotte; van der Stam, Ward; Duchoň, Tomáš; Šmíd, Břetislav; Groeneveld, Esther; Berben, Peter; Bals, Sara; Weckhuysen, Bert M.
Restructuring of titanium oxide overlayers over nickel nanoparticles during catalysis Journal Article
In: Science, vol. 380, no. 6645, pp. 644 – 651, 2023, (Cited by: 27).
@article{Monai2023644,
title = {Restructuring of titanium oxide overlayers over nickel nanoparticles during catalysis},
author = { Matteo Monai and Kellie Jenkinson and Angela E.M. Melcherts and Jaap N. Louwen and Ece A. Irmak and Sandra Van Aert and Thomas Altantzis and Charlotte Vogt and Ward van der Stam and Tomáš Duchoň and Břetislav Šmíd and Esther Groeneveld and Peter Berben and Sara Bals and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85159739034&doi=10.1126%2fSCIENCE.ADF6984&partnerID=40&md5=c248c10431a229396864af481e47f032},
doi = {10.1126/SCIENCE.ADF6984},
year = {2023},
date = {2023-01-01},
journal = {Science},
volume = {380},
number = {6645},
pages = {644 – 651},
note = {Cited by: 27},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Ding, Pan; An, Hongyu; Zellner, Philipp; Guan, Tianfu; Gao, Jianyong; Müller-Buschbaum, Peter; Weckhuysen, Bert M.; van der Stam, Ward; Sharp, Ian D.
Elucidating the Roles of Nafion/Solvent Formulations in Copper-Catalyzed CO2 Electrolysis Journal Article
In: ACS Catalysis, vol. 13, no. 8, pp. 5336 – 5347, 2023, (Cited by: 7; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Ding20235336,
title = {Elucidating the Roles of Nafion/Solvent Formulations in Copper-Catalyzed CO2 Electrolysis},
author = { Pan Ding and Hongyu An and Philipp Zellner and Tianfu Guan and Jianyong Gao and Peter Müller-Buschbaum and Bert M. Weckhuysen and Ward van der Stam and Ian D. Sharp},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85152200375&doi=10.1021%2facscatal.2c05235&partnerID=40&md5=86c1330281cc2037a6340fb5cb882a1a},
doi = {10.1021/acscatal.2c05235},
year = {2023},
date = {2023-01-01},
journal = {ACS Catalysis},
volume = {13},
number = {8},
pages = {5336 – 5347},
note = {Cited by: 7; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Couce, Pedro Mazaira; Madsen, Thor Kongstad; Plaza-Mayoral, Elena; Kristoffersen, Henrik H.; Chorkendorff, Ib; Dalby, Kim Nicole; van der Stam, Ward; Rossmeisl, Jan; Escudero-Escribano, María; Sebastián-Pascual, Paula
Tailoring the facet distribution on copper with chloride Journal Article
In: Chemical Science, vol. 15, no. 5, pp. 1714 – 1725, 2023, (Cited by: 0; All Open Access, Gold Open Access).
@article{Couce20231714,
title = {Tailoring the facet distribution on copper with chloride},
author = { Pedro Mazaira Couce and Thor Kongstad Madsen and Elena Plaza-Mayoral and Henrik H. Kristoffersen and Ib Chorkendorff and Kim Nicole Dalby and Ward van der Stam and Jan Rossmeisl and María Escudero-Escribano and Paula Sebastián-Pascual},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85181481045&doi=10.1039%2fd3sc05988j&partnerID=40&md5=afb2703d093bc8acb57a20e9918901c6},
doi = {10.1039/d3sc05988j},
year = {2023},
date = {2023-01-01},
journal = {Chemical Science},
volume = {15},
number = {5},
pages = {1714 – 1725},
note = {Cited by: 0; All Open Access, Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
An, Hongyu; de Ruiter, Jim; Wu, Longfei; Yang, Shuang; Meirer, Florian; van der Stam, Ward; Weckhuysen, Bert M.
Spatiotemporal Mapping of Local Heterogeneities during Electrochemical Carbon Dioxide Reduction Journal Article
In: JACS Au, vol. 3, no. 7, pp. 1890 – 1901, 2023, (Cited by: 1; All Open Access, Gold Open Access, Green Open Access).
@article{An20231890,
title = {Spatiotemporal Mapping of Local Heterogeneities during Electrochemical Carbon Dioxide Reduction},
author = { Hongyu An and Jim de Ruiter and Longfei Wu and Shuang Yang and Florian Meirer and Ward van der Stam and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85164952305&doi=10.1021%2fjacsau.3c00129&partnerID=40&md5=461599b507b7271665dc772a9118f349},
doi = {10.1021/jacsau.3c00129},
year = {2023},
date = {2023-01-01},
journal = {JACS Au},
volume = {3},
number = {7},
pages = {1890 – 1901},
note = {Cited by: 1; All Open Access, Gold Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W. Van Der
The Necessity for Multiscale In Situ Characterization of Tailored Electrocatalyst Nanoparticle Stability † Journal Article
In: Chemistry of Materials, 2022, (cited By 0).
@article{VanDerStam2022,
title = {The Necessity for Multiscale In Situ Characterization of Tailored Electrocatalyst Nanoparticle Stability †},
author = { W. Van Der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85145991987&doi=10.1021%2facs.chemmater.2c03286&partnerID=40&md5=b3bf2af3ec65a648b558fabba26884be},
doi = {10.1021/acs.chemmater.2c03286},
year = {2022},
date = {2022-01-01},
journal = {Chemistry of Materials},
note = {cited By 0},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Yang, Shuang; Liu, Zhaochun; An, Hongyu; Arnouts, Sven; Ruiter, Jim De; Rollier, Floriane; Bals, Sara; Altantzis, Thomas; Figueiredo, Marta C.; Filot, Ivo A. W.; Hensen, Emiel J. M.; Weckhuysen, Bert M.; Stam, Ward Van Der
Near-Unity Electrochemical CO2to CO Conversion over Sn-Doped Copper Oxide Nanoparticles Journal Article
In: ACS Catalysis, vol. 12, no. 24, pp. 15146 – 15156, 2022, (Cited by: 14; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Yang202215146,
title = {Near-Unity Electrochemical CO2to CO Conversion over Sn-Doped Copper Oxide Nanoparticles},
author = { Shuang Yang and Zhaochun Liu and Hongyu An and Sven Arnouts and Jim De Ruiter and Floriane Rollier and Sara Bals and Thomas Altantzis and Marta C. Figueiredo and Ivo A.W. Filot and Emiel J.M. Hensen and Bert M. Weckhuysen and Ward Van Der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85143434989&doi=10.1021%2facscatal.2c04279&partnerID=40&md5=574727e81bacac9e7703629ca2c28476},
doi = {10.1021/acscatal.2c04279},
year = {2022},
date = {2022-01-01},
journal = {ACS Catalysis},
volume = {12},
number = {24},
pages = {15146 – 15156},
note = {Cited by: 14; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Ruiter, Jim De; An, Hongyu; Wu, Longfei; Gijsberg, Zamorano; Yang, Shuang; Hartman, Thomas; Weckhuysen, Bert M.; Stam, Ward Van Der
Probing the Dynamics of Low-Overpotential CO2-to-CO Activation on Copper Electrodes with Time-Resolved Raman Spectroscopy Journal Article
In: Journal of the American Chemical Society, vol. 144, no. 33, pp. 15047 – 15058, 2022, (Cited by: 32; All Open Access, Green Open Access).
@article{DeRuiter202215047b,
title = {Probing the Dynamics of Low-Overpotential CO2-to-CO Activation on Copper Electrodes with Time-Resolved Raman Spectroscopy},
author = { Jim De Ruiter and Hongyu An and Longfei Wu and Zamorano Gijsberg and Shuang Yang and Thomas Hartman and Bert M. Weckhuysen and Ward Van Der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85136895795&doi=10.1021%2fjacs.2c03172&partnerID=40&md5=0d782eb225f2788259027a821deee3dc},
doi = {10.1021/jacs.2c03172},
year = {2022},
date = {2022-01-01},
journal = {Journal of the American Chemical Society},
volume = {144},
number = {33},
pages = {15047 – 15058},
note = {Cited by: 32; All Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Yang, Shuang; An, Hongyu; Anastasiadou, Dimitra; Xu, Wenjie; Wu, Longfei; Wang, Hui; de Ruiter, Jim; Arnouts, Sven; Figueiredo, Marta C.; Bals, Sara; Altantzis, Thomas; van der Stam, Ward; Weckhuysen, Bert M.
Waste-Derived Copper-Lead Electrocatalysts for CO2 Reduction Journal Article
In: ChemCatChem, vol. 14, no. 18, 2022, (Cited by: 7; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Yang2022,
title = {Waste-Derived Copper-Lead Electrocatalysts for CO2 Reduction},
author = { Shuang Yang and Hongyu An and Dimitra Anastasiadou and Wenjie Xu and Longfei Wu and Hui Wang and Jim de Ruiter and Sven Arnouts and Marta C. Figueiredo and Sara Bals and Thomas Altantzis and Ward van der Stam and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85134312014&doi=10.1002%2fcctc.202200754&partnerID=40&md5=34381883fafc53ff9986154fdbbf6215},
doi = {10.1002/cctc.202200754},
year = {2022},
date = {2022-01-01},
journal = {ChemCatChem},
volume = {14},
number = {18},
note = {Cited by: 7; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der
The Necessity for Multiscale In Situ Characterization of Tailored Electrocatalyst Nanoparticle Stability † Journal Article
In: Chemistry of Materials, 2022, (Cited by: 1; All Open Access, Hybrid Gold Open Access).
@article{VanDerStam2022b,
title = {The Necessity for Multiscale In Situ Characterization of Tailored Electrocatalyst Nanoparticle Stability †},
author = { Ward Van Der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85145991987&doi=10.1021%2facs.chemmater.2c03286&partnerID=40&md5=b3bf2af3ec65a648b558fabba26884be},
doi = {10.1021/acs.chemmater.2c03286},
year = {2022},
date = {2022-01-01},
journal = {Chemistry of Materials},
note = {Cited by: 1; All Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
An, Hongyu; Wu, Longfei; Mandemaker, Laurens D. B.; Yang, Shuang; de Ruiter, Jim; Wijten, Jochem H. J.; Janssens, Joris C. L.; Hartman, Thomas; van der Stam, Ward; Weckhuysen, Bert M.
Sub-Second Time-Resolved Surface-Enhanced Raman Spectroscopy Reveals Dynamic CO Intermediates during Electrochemical CO2 Reduction on Copper Journal Article
In: Angewandte Chemie - International Edition, vol. 60, no. 30, pp. 16576 – 16584, 2021, (Cited by: 125; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{An202116576,
title = {Sub-Second Time-Resolved Surface-Enhanced Raman Spectroscopy Reveals Dynamic CO Intermediates during Electrochemical CO2 Reduction on Copper},
author = { Hongyu An and Longfei Wu and Laurens D. B. Mandemaker and Shuang Yang and Jim de Ruiter and Jochem H. J. Wijten and Joris C. L. Janssens and Thomas Hartman and Ward van der Stam and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85107959879&doi=10.1002%2fanie.202104114&partnerID=40&md5=14ec7cc47637cc245a2539b7c8664d15},
doi = {10.1002/anie.202104114},
year = {2021},
date = {2021-01-01},
journal = {Angewandte Chemie - International Edition},
volume = {60},
number = {30},
pages = {16576 – 16584},
note = {Cited by: 125; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Wu, Longfei; Kolmeijer, Kees E.; Zhang, Yue; An, Hongyu; Arnouts, Sven; Bals, Sara; Altantzis, Thomas; Hofmann, Jan P.; Figueiredo, Marta Costa; Hensen, Emiel J. M.; Weckhuysen, Bert M.; Stam, Ward Van Der
Stabilization effects in binary colloidal Cu and Ag nanoparticle electrodes under electrochemical CO2reduction conditions Journal Article
In: Nanoscale, vol. 13, no. 9, pp. 4835 – 4844, 2021, (Cited by: 24; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Wu20214835,
title = {Stabilization effects in binary colloidal Cu and Ag nanoparticle electrodes under electrochemical CO2reduction conditions},
author = { Longfei Wu and Kees E. Kolmeijer and Yue Zhang and Hongyu An and Sven Arnouts and Sara Bals and Thomas Altantzis and Jan P. Hofmann and Marta Costa Figueiredo and Emiel J. M. Hensen and Bert M. Weckhuysen and Ward Van Der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85102415523&doi=10.1039%2fd0nr09040a&partnerID=40&md5=5196f833d12448f745658ba15739b5f0},
doi = {10.1039/d0nr09040a},
year = {2021},
date = {2021-01-01},
journal = {Nanoscale},
volume = {13},
number = {9},
pages = {4835 – 4844},
note = {Cited by: 24; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Hartman, T; Geitenbeek, R G; Wondergem, C S; Stam, W Van Der; Weckhuysen, B M
Operando Nanoscale Sensors in Catalysis: All Eyes on Catalyst Particles Journal Article
In: ACS Nano, 2020, (cited By 0).
@article{Hartman2020,
title = {Operando Nanoscale Sensors in Catalysis: All Eyes on Catalyst Particles},
author = {T Hartman and R G Geitenbeek and C S Wondergem and W Van Der Stam and B M Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85083881076&doi=10.1021%2facsnano.9b09834&partnerID=40&md5=02c08f4d6d127819f2e8a6db4df5cfdd},
doi = {10.1021/acsnano.9b09834},
year = {2020},
date = {2020-01-01},
journal = {ACS Nano},
note = {cited By 0},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Hartman, T; Geitenbeek, R G; Wondergem, C S; Stam, W Van Der; Weckhuysen, B M
Operando Nanoscale Sensors in Catalysis: All Eyes on Catalyst Particles Journal Article
In: ACS Nano, 2020, (cited By 9).
@article{Hartman2020b,
title = {Operando Nanoscale Sensors in Catalysis: All Eyes on Catalyst Particles},
author = {T Hartman and R G Geitenbeek and C S Wondergem and W Van Der Stam and B M Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85083881076&doi=10.1021%2facsnano.9b09834&partnerID=40&md5=02c08f4d6d127819f2e8a6db4df5cfdd},
doi = {10.1021/acsnano.9b09834},
year = {2020},
date = {2020-01-01},
journal = {ACS Nano},
note = {cited By 9},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Gélvez-Rueda, María C.; Fridriksson, Magnus B.; Dubey, Rajeev K.; Jager, Wolter F.; van der Stam, Ward; Grozema, Ferdinand C.
Overcoming the exciton binding energy in two-dimensional perovskite nanoplatelets by attachment of conjugated organic chromophores Journal Article
In: Nature Communications, vol. 11, no. 1, 2020, (Cited by: 79; All Open Access, Gold Open Access, Green Open Access).
@article{Gélvez-Rueda2020,
title = {Overcoming the exciton binding energy in two-dimensional perovskite nanoplatelets by attachment of conjugated organic chromophores},
author = { María C. Gélvez-Rueda and Magnus B. Fridriksson and Rajeev K. Dubey and Wolter F. Jager and Ward van der Stam and Ferdinand C. Grozema},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85083798821&doi=10.1038%2fs41467-020-15869-7&partnerID=40&md5=dbe73b993adf58b813715ce8e62254cc},
doi = {10.1038/s41467-020-15869-7},
year = {2020},
date = {2020-01-01},
journal = {Nature Communications},
volume = {11},
number = {1},
note = {Cited by: 79; All Open Access, Gold Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Vonk, Sander J. W.; Fridriksson, Magnus B.; Hinterding, Stijn O. M.; Mangnus, Mark J. J.; Swieten, Thomas P. Van; Grozema, Ferdinand C.; Rabouw, Freddy T.; Stam, Ward Van Der
Trapping and Detrapping in Colloidal Perovskite Nanoplatelets: Elucidation and Prevention of Nonradiative Processes through Chemical Treatment Journal Article
In: Journal of Physical Chemistry C, vol. 124, no. 14, pp. 8047 – 8054, 2020, (Cited by: 21; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Vonk20208047,
title = {Trapping and Detrapping in Colloidal Perovskite Nanoplatelets: Elucidation and Prevention of Nonradiative Processes through Chemical Treatment},
author = { Sander J. W. Vonk and Magnus B. Fridriksson and Stijn O. M. Hinterding and Mark J. J. Mangnus and Thomas P. Van Swieten and Ferdinand C. Grozema and Freddy T. Rabouw and Ward Van Der Stam},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85084036259&doi=10.1021%2facs.jpcc.0c02287&partnerID=40&md5=b2a928d6323282f6c98ee3fd8d84f0d6},
doi = {10.1021/acs.jpcc.0c02287},
year = {2020},
date = {2020-01-01},
journal = {Journal of Physical Chemistry C},
volume = {124},
number = {14},
pages = {8047 – 8054},
note = {Cited by: 21; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Hartman, Thomas; Geitenbeek, Robin G.; Wondergem, Caterina S.; van der Stam, Ward; Weckhuysen, Bert M.
Operando Nanoscale Sensors in Catalysis: All Eyes on Catalyst Particles Journal Article
In: ACS Nano, vol. 14, no. 4, pp. 3725 – 3735, 2020, (Cited by: 54; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Hartman20203725,
title = {Operando Nanoscale Sensors in Catalysis: All Eyes on Catalyst Particles},
author = { Thomas Hartman and Robin G. Geitenbeek and Caterina S. Wondergem and Ward van der Stam and Bert M. Weckhuysen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85083881076&doi=10.1021%2facsnano.9b09834&partnerID=40&md5=02c08f4d6d127819f2e8a6db4df5cfdd},
doi = {10.1021/acsnano.9b09834},
year = {2020},
date = {2020-01-01},
journal = {ACS Nano},
volume = {14},
number = {4},
pages = {3725 – 3735},
note = {Cited by: 54; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Grimaldi, G; Geuchies, J J; Gudjonsdottir, S; Uffelen, P T Van; Overeem, M Van; Brynjarsson, B; Kirkwood, N; Houtepen, A J
Electrochemical modulation of the photophysics of surface-localized trap states in core/shell/(shell) quantum dot films Journal Article
In: Chemistry of Materials, vol. 31, no. 20, pp. 8484-8493, 2019, (cited By 8).
@article{VanDerStam20198484,
title = {Electrochemical modulation of the photophysics of surface-localized trap states in core/shell/(shell) quantum dot films},
author = {W Van Der Stam and G Grimaldi and J J Geuchies and S Gudjonsdottir and P T Van Uffelen and M Van Overeem and B Brynjarsson and N Kirkwood and A J Houtepen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85073056706&doi=10.1021%2facs.chemmater.9b02908&partnerID=40&md5=47e45063307dd0a7ba2bece72795fa73},
doi = {10.1021/acs.chemmater.9b02908},
year = {2019},
date = {2019-01-01},
journal = {Chemistry of Materials},
volume = {31},
number = {20},
pages = {8484-8493},
note = {cited By 8},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Hinterding, Stijn O. M.; Berends, Anne C.; Kurttepeli, Mert; Moret, Marc-Etienne; Meeldijk, Johannes D.; Bals, Sara; Stam, Ward Van Der; Donega, Celso De Mello
Tailoring Cu+ for Ga3+ Cation Exchange in Cu2- xS and CuInS2 Nanocrystals by Controlling the Ga Precursor Chemistry Journal Article
In: ACS Nano, vol. 13, no. 11, pp. 12880 – 12893, 2019, (Cited by: 28; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Hinterding201912880,
title = {Tailoring Cu+ for Ga3+ Cation Exchange in Cu2- xS and CuInS2 Nanocrystals by Controlling the Ga Precursor Chemistry},
author = { Stijn O. M. Hinterding and Anne C. Berends and Mert Kurttepeli and Marc-Etienne Moret and Johannes D. Meeldijk and Sara Bals and Ward Van Der Stam and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85074253775&doi=10.1021%2facsnano.9b05337&partnerID=40&md5=c8702a552ab82a9a799b498bcc4bdb16},
doi = {10.1021/acsnano.9b05337},
year = {2019},
date = {2019-01-01},
journal = {ACS Nano},
volume = {13},
number = {11},
pages = {12880 – 12893},
note = {Cited by: 28; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Grimaldi, Gianluca; Geuchies, Jaco J.; Gudjonsdottir, Solrun; Uffelen, Pieter T. Van; Overeem, Mandy Van; Brynjarsson, Baldur; Kirkwood, Nicholas; Houtepen, Arjan J.
Electrochemical modulation of the photophysics of surface-localized trap states in core/shell/(shell) quantum dot films Journal Article
In: Chemistry of Materials, vol. 31, no. 20, pp. 8484 – 8493, 2019, (Cited by: 31; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{VanDerStam20198484b,
title = {Electrochemical modulation of the photophysics of surface-localized trap states in core/shell/(shell) quantum dot films},
author = { Ward Van Der Stam and Gianluca Grimaldi and Jaco J. Geuchies and Solrun Gudjonsdottir and Pieter T. Van Uffelen and Mandy Van Overeem and Baldur Brynjarsson and Nicholas Kirkwood and Arjan J. Houtepen},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85073056706&doi=10.1021%2facs.chemmater.9b02908&partnerID=40&md5=47e45063307dd0a7ba2bece72795fa73},
doi = {10.1021/acs.chemmater.9b02908},
year = {2019},
date = {2019-01-01},
journal = {Chemistry of Materials},
volume = {31},
number = {20},
pages = {8484 – 8493},
note = {Cited by: 31; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Berends, Anne C.; Stam, Ward Van Der; Akkerman, Quinten A.; Meeldijk, Johannes D.; Lit, Joost Van Der; Donega, Celso De Mello
Anisotropic 2D Cu2- xSe Nanocrystals from Dodecaneselenol and Their Conversion to CdSe and CuInSe2 Nanoparticles Journal Article
In: Chemistry of Materials, vol. 30, no. 11, pp. 3836 – 3846, 2018, (Cited by: 24; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Berends20183836,
title = {Anisotropic 2D Cu2- xSe Nanocrystals from Dodecaneselenol and Their Conversion to CdSe and CuInSe2 Nanoparticles},
author = { Anne C. Berends and Ward Van Der Stam and Quinten A. Akkerman and Johannes D. Meeldijk and Joost Van Der Lit and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85046962415&doi=10.1021%2facs.chemmater.8b01143&partnerID=40&md5=06d2989664ccbdc91dc9ea4034d93348},
doi = {10.1021/acs.chemmater.8b01143},
year = {2018},
date = {2018-01-01},
journal = {Chemistry of Materials},
volume = {30},
number = {11},
pages = {3836 – 3846},
note = {Cited by: 24; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Berends, Anne C.; Stam, Ward Van Der; Hofmann, Jan P.; Bladt, Eva; Meeldijk, Johannes D.; Bals, Sara; Donega, Celso De Mello
Interplay between Surface Chemistry, Precursor Reactivity, and Temperature Determines Outcome of ZnS Shelling Reactions on CuInS2 Nanocrystals Journal Article
In: Chemistry of Materials, vol. 30, no. 7, pp. 2400 – 2413, 2018, (Cited by: 83; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{Berends20182400,
title = {Interplay between Surface Chemistry, Precursor Reactivity, and Temperature Determines Outcome of ZnS Shelling Reactions on CuInS2 Nanocrystals},
author = { Anne C. Berends and Ward Van Der Stam and Jan P. Hofmann and Eva Bladt and Johannes D. Meeldijk and Sara Bals and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85045190366&doi=10.1021%2facs.chemmater.8b00477&partnerID=40&md5=a371782d6d9a9cbd825ff404ea4f7506},
doi = {10.1021/acs.chemmater.8b00477},
year = {2018},
date = {2018-01-01},
journal = {Chemistry of Materials},
volume = {30},
number = {7},
pages = {2400 – 2413},
note = {Cited by: 83; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
der Stam, W Van; Geuchies, J J; Altantzis, T; Bos, K H W Van Den; Meeldijk, J D; Aert, S Van; Bals, S; Vanmaekelbergh, D; Donega, C De Mello
Highly Emissive Divalent-Ion-Doped Colloidal CsPb1-xMxBr3 Perovskite Nanocrystals through Cation Exchange Journal Article
In: Journal of the American Chemical Society, vol. 139, no. 11, pp. 4087-4097, 2017, (cited By 325).
@article{VanderStam20174087,
title = {Highly Emissive Divalent-Ion-Doped Colloidal CsPb1-xMxBr3 Perovskite Nanocrystals through Cation Exchange},
author = {W Van der Stam and J J Geuchies and T Altantzis and K H W Van Den Bos and J D Meeldijk and S Van Aert and S Bals and D Vanmaekelbergh and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85015873930&doi=10.1021%2fjacs.6b13079&partnerID=40&md5=c1a15a584bdc7d13cd9c9ff8f30c21d6},
doi = {10.1021/jacs.6b13079},
year = {2017},
date = {2017-01-01},
journal = {Journal of the American Chemical Society},
volume = {139},
number = {11},
pages = {4087-4097},
note = {cited By 325},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
der Stam, Ward Van; Geuchies, Jaco J.; Altantzis, Thomas; Bos, Karel H. W. Van Den; Meeldijk, Johannes D.; Aert, Sandra Van; Bals, Sara; Vanmaekelbergh, Daniel; Donega, Celso De Mello
Highly Emissive Divalent-Ion-Doped Colloidal CsPb1-xMxBr3 Perovskite Nanocrystals through Cation Exchange Journal Article
In: Journal of the American Chemical Society, vol. 139, no. 11, pp. 4087 – 4097, 2017, (Cited by: 572; All Open Access, Green Open Access, Hybrid Gold Open Access).
@article{VanderStam20174087b,
title = {Highly Emissive Divalent-Ion-Doped Colloidal CsPb1-xMxBr3 Perovskite Nanocrystals through Cation Exchange},
author = { Ward Van der Stam and Jaco J. Geuchies and Thomas Altantzis and Karel H. W. Van Den Bos and Johannes D. Meeldijk and Sandra Van Aert and Sara Bals and Daniel Vanmaekelbergh and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-85015873930&doi=10.1021%2fjacs.6b13079&partnerID=40&md5=c1a15a584bdc7d13cd9c9ff8f30c21d6},
doi = {10.1021/jacs.6b13079},
year = {2017},
date = {2017-01-01},
journal = {Journal of the American Chemical Society},
volume = {139},
number = {11},
pages = {4087 – 4097},
note = {Cited by: 572; All Open Access, Green Open Access, Hybrid Gold Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Gradmann, S; Altantzis, T; Ke, X; Baldus, M; Bals, S; Donega, C De Mello
Shape control of colloidal Cu2-xS polyhedral nanocrystals by tuning the nucleation rates Journal Article
In: Chemistry of Materials, vol. 28, no. 18, pp. 6705-6715, 2016, (cited By 22).
@article{VanDerStam20166705,
title = {Shape control of colloidal Cu2-xS polyhedral nanocrystals by tuning the nucleation rates},
author = {W Van Der Stam and S Gradmann and T Altantzis and X Ke and M Baldus and S Bals and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84989172561&doi=10.1021%2facs.chemmater.6b03098&partnerID=40&md5=dbdd422749f0afe5db55e2ed2ef065ca},
doi = {10.1021/acs.chemmater.6b03098},
year = {2016},
date = {2016-01-01},
journal = {Chemistry of Materials},
volume = {28},
number = {18},
pages = {6705-6715},
note = {cited By 22},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Rabouw, F T; Geuchies, J J; Berends, A C; Hinterding, S O M; Geitenbeek, R G; Lit, J Van Der; Prévost, S; Petukhov, A V; Donega, C De Mello
In Situ Probing of Stack-Templated Growth of Ultrathin Cu2-xS Nanosheets Journal Article
In: Chemistry of Materials, vol. 28, no. 17, pp. 6381-6389, 2016, (cited By 14).
@article{VanDerStam20166381,
title = {In Situ Probing of Stack-Templated Growth of Ultrathin Cu2-xS Nanosheets},
author = {W Van Der Stam and F T Rabouw and J J Geuchies and A C Berends and S O M Hinterding and R G Geitenbeek and J Van Der Lit and S Prévost and A V Petukhov and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84987606049&doi=10.1021%2facs.chemmater.6b02787&partnerID=40&md5=acc18537151ccf44a1ac6f189e964c92},
doi = {10.1021/acs.chemmater.6b02787},
year = {2016},
date = {2016-01-01},
journal = {Chemistry of Materials},
volume = {28},
number = {17},
pages = {6381-6389},
note = {cited By 14},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Rabouw, F T; Vonk, S J W; Geuchies, J J; Ligthart, H; Petukhov, A V; Donega, C De Mello
Oleic Acid-Induced Atomic Alignment of ZnS Polyhedral Nanocrystals Journal Article
In: Nano Letters, vol. 16, no. 4, pp. 2608-2614, 2016, (cited By 29).
@article{VanDerStam20162608,
title = {Oleic Acid-Induced Atomic Alignment of ZnS Polyhedral Nanocrystals},
author = {W Van Der Stam and F T Rabouw and S J W Vonk and J J Geuchies and H Ligthart and A V Petukhov and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84964869566&doi=10.1021%2facs.nanolett.6b00221&partnerID=40&md5=470218ce6e61e0b6109cf0a69e5153de},
doi = {10.1021/acs.nanolett.6b00221},
year = {2016},
date = {2016-01-01},
journal = {Nano Letters},
volume = {16},
number = {4},
pages = {2608-2614},
note = {cited By 29},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Berends, A C; Donega, C De Mello
Prospects of Colloidal Copper Chalcogenide Nanocrystals Journal Article
In: ChemPhysChem, vol. 17, no. 5, pp. 559-581, 2016, (cited By 87).
@article{VanDerStam2016559,
title = {Prospects of Colloidal Copper Chalcogenide Nanocrystals},
author = {W Van Der Stam and A C Berends and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84960337499&doi=10.1002%2fcphc.201500976&partnerID=40&md5=3c7037e5d2aa900799c60ab4c5dd0122},
doi = {10.1002/cphc.201500976},
year = {2016},
date = {2016-01-01},
journal = {ChemPhysChem},
volume = {17},
number = {5},
pages = {559-581},
note = {cited By 87},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Rabouw, F T; Cogan, N M B; Berends, A C; Stam, W V D; Vanmaekelbergh, D; Koenderink, A F; Krauss, T D; Donega, C D M
Non-blinking single-photon emitters in silica Journal Article
In: Scientific Reports, vol. 6, 2016, (cited By 18).
@article{Rabouw2016c,
title = {Non-blinking single-photon emitters in silica},
author = {F T Rabouw and N M B Cogan and A C Berends and W V D Stam and D Vanmaekelbergh and A F Koenderink and T D Krauss and C D M Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84959020156&doi=10.1038%2fsrep21187&partnerID=40&md5=24a37a78be9badb85641dfe5af35b8ae},
doi = {10.1038/srep21187},
year = {2016},
date = {2016-01-01},
journal = {Scientific Reports},
volume = {6},
note = {cited By 18},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Rabouw, Freddy T.; Vonk, Sander J. W.; Geuchies, Jaco J.; Ligthart, Hans; Petukhov, Andrei V.; Donega, Celso De Mello
Oleic Acid-Induced Atomic Alignment of ZnS Polyhedral Nanocrystals Journal Article
In: Nano Letters, vol. 16, no. 4, pp. 2608 – 2614, 2016, (Cited by: 32; All Open Access, Bronze Open Access, Green Open Access).
@article{VanDerStam20162608b,
title = {Oleic Acid-Induced Atomic Alignment of ZnS Polyhedral Nanocrystals},
author = { Ward Van Der Stam and Freddy T. Rabouw and Sander J. W. Vonk and Jaco J. Geuchies and Hans Ligthart and Andrei V. Petukhov and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84964869566&doi=10.1021%2facs.nanolett.6b00221&partnerID=40&md5=470218ce6e61e0b6109cf0a69e5153de},
doi = {10.1021/acs.nanolett.6b00221},
year = {2016},
date = {2016-01-01},
journal = {Nano Letters},
volume = {16},
number = {4},
pages = {2608 – 2614},
note = {Cited by: 32; All Open Access, Bronze Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Gradmann, Sabine; Altantzis, Thomas; Ke, Xiaoxing; Baldus, Marc; Bals, Sara; Donega, Celso De Mello
Shape control of colloidal Cu2-xS polyhedral nanocrystals by tuning the nucleation rates Journal Article
In: Chemistry of Materials, vol. 28, no. 18, pp. 6705 – 6715, 2016, (Cited by: 26; All Open Access, Bronze Open Access, Green Open Access).
@article{VanDerStam20166705b,
title = {Shape control of colloidal Cu2-xS polyhedral nanocrystals by tuning the nucleation rates},
author = { Ward Van Der Stam and Sabine Gradmann and Thomas Altantzis and Xiaoxing Ke and Marc Baldus and Sara Bals and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84989172561&doi=10.1021%2facs.chemmater.6b03098&partnerID=40&md5=dbdd422749f0afe5db55e2ed2ef065ca},
doi = {10.1021/acs.chemmater.6b03098},
year = {2016},
date = {2016-01-01},
journal = {Chemistry of Materials},
volume = {28},
number = {18},
pages = {6705 – 6715},
note = {Cited by: 26; All Open Access, Bronze Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Berends, Anne C.; Donega, Celso De Mello
Prospects of Colloidal Copper Chalcogenide Nanocrystals Journal Article
In: ChemPhysChem, vol. 17, no. 5, pp. 559 – 581, 2016, (Cited by: 124; All Open Access, Green Open Access).
@article{VanDerStam2016559b,
title = {Prospects of Colloidal Copper Chalcogenide Nanocrystals},
author = { Ward Van Der Stam and Anne C. Berends and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84960337499&doi=10.1002%2fcphc.201500976&partnerID=40&md5=3c7037e5d2aa900799c60ab4c5dd0122},
doi = {10.1002/cphc.201500976},
year = {2016},
date = {2016-01-01},
journal = {ChemPhysChem},
volume = {17},
number = {5},
pages = {559 – 581},
note = {Cited by: 124; All Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Rabouw, Freddy T.; Geuchies, Jaco J.; Berends, Anne C.; Hinterding, Stijn O. M.; Geitenbeek, Robin G.; Lit, Joost Van Der; Prévost, Sylvain; Petukhov, Andrei V.; Donega, Celso De Mello
In Situ Probing of Stack-Templated Growth of Ultrathin Cu2-xS Nanosheets Journal Article
In: Chemistry of Materials, vol. 28, no. 17, pp. 6381 – 6389, 2016, (Cited by: 27; All Open Access, Bronze Open Access, Green Open Access).
@article{VanDerStam20166381b,
title = {In Situ Probing of Stack-Templated Growth of Ultrathin Cu2-xS Nanosheets},
author = { Ward Van Der Stam and Freddy T. Rabouw and Jaco J. Geuchies and Anne C. Berends and Stijn O.M. Hinterding and Robin G. Geitenbeek and Joost Van Der Lit and Sylvain Prévost and Andrei V. Petukhov and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84987606049&doi=10.1021%2facs.chemmater.6b02787&partnerID=40&md5=acc18537151ccf44a1ac6f189e964c92},
doi = {10.1021/acs.chemmater.6b02787},
year = {2016},
date = {2016-01-01},
journal = {Chemistry of Materials},
volume = {28},
number = {17},
pages = {6381 – 6389},
note = {Cited by: 27; All Open Access, Bronze Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Bladt, E; Rabouw, F T; Bals, S; Donega, C De Mello
Near-Infrared Emitting CuInSe2/CuInS2 Dot Core/Rod Shell Heteronanorods by Sequential Cation Exchange Journal Article
In: ACS Nano, vol. 9, no. 11, pp. 11430-11438, 2015, (cited By 66).
@article{VanDerStam201511430,
title = {Near-Infrared Emitting CuInSe2/CuInS2 Dot Core/Rod Shell Heteronanorods by Sequential Cation Exchange},
author = {W Van Der Stam and E Bladt and F T Rabouw and S Bals and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84948414454&doi=10.1021%2facsnano.5b05496&partnerID=40&md5=599559d79c1629fd6ebce6e1356d219d},
doi = {10.1021/acsnano.5b05496},
year = {2015},
date = {2015-01-01},
journal = {ACS Nano},
volume = {9},
number = {11},
pages = {11430-11438},
note = {cited By 66},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Berends, A C; Rabouw, F T; Willhammar, T; Ke, X; Meeldijk, J D; Bals, S; Donega, C De Mello
Luminescent CuInS2 quantum dots by partial cation exchange in Cu2- xS nanocrystals Journal Article
In: Chemistry of Materials, vol. 27, no. 2, pp. 621-628, 2015, (cited By 93).
@article{VanDerStam2015621,
title = {Luminescent CuInS2 quantum dots by partial cation exchange in Cu2- xS nanocrystals},
author = {W Van Der Stam and A C Berends and F T Rabouw and T Willhammar and X Ke and J D Meeldijk and S Bals and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84921731109&doi=10.1021%2fcm504340h&partnerID=40&md5=c6a111289f1683b897c4549634cc0919},
doi = {10.1021/cm504340h},
year = {2015},
date = {2015-01-01},
journal = {Chemistry of Materials},
volume = {27},
number = {2},
pages = {621-628},
note = {cited By 93},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Akkerman, Q A; Ke, X; Huis, M A Van; Bals, S; Donega, C De Mello
Solution-processable ultrathin size- and shape-controlled colloidal Cu2- xS nanosheets Journal Article
In: Chemistry of Materials, vol. 27, no. 1, pp. 283-291, 2015, (cited By 59).
@article{VanDerStam2015283,
title = {Solution-processable ultrathin size- and shape-controlled colloidal Cu2- xS nanosheets},
author = {W Van Der Stam and Q A Akkerman and X Ke and M A Van Huis and S Bals and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84921348715&doi=10.1021%2fcm503929q&partnerID=40&md5=22b8b3fe5d0c2bb8f9916861ffbbc219},
doi = {10.1021/cm503929q},
year = {2015},
date = {2015-01-01},
journal = {Chemistry of Materials},
volume = {27},
number = {1},
pages = {283-291},
note = {cited By 59},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Bladt, Eva; Rabouw, Freddy T.; Bals, Sara; Donega, Celso De Mello
Near-Infrared Emitting CuInSe2/CuInS2 Dot Core/Rod Shell Heteronanorods by Sequential Cation Exchange Journal Article
In: ACS Nano, vol. 9, no. 11, pp. 11430 – 11438, 2015, (Cited by: 98; All Open Access, Bronze Open Access, Green Open Access).
@article{VanDerStam201511430b,
title = {Near-Infrared Emitting CuInSe2/CuInS2 Dot Core/Rod Shell Heteronanorods by Sequential Cation Exchange},
author = { Ward Van Der Stam and Eva Bladt and Freddy T. Rabouw and Sara Bals and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84948414454&doi=10.1021%2facsnano.5b05496&partnerID=40&md5=599559d79c1629fd6ebce6e1356d219d},
doi = {10.1021/acsnano.5b05496},
year = {2015},
date = {2015-01-01},
journal = {ACS Nano},
volume = {9},
number = {11},
pages = {11430 – 11438},
note = {Cited by: 98; All Open Access, Bronze Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Berends, Anne C.; Rabouw, Freddy T.; Willhammar, Tom; Ke, Xiaoxing; Meeldijk, Johannes D.; Bals, Sara; Donega, Celso De Mello
Luminescent CuInS2 quantum dots by partial cation exchange in Cu2- xS nanocrystals Journal Article
In: Chemistry of Materials, vol. 27, no. 2, pp. 621 – 628, 2015, (Cited by: 122; All Open Access, Green Open Access).
@article{VanDerStam2015621b,
title = {Luminescent CuInS2 quantum dots by partial cation exchange in Cu2- xS nanocrystals},
author = { Ward Van Der Stam and Anne C. Berends and Freddy T. Rabouw and Tom Willhammar and Xiaoxing Ke and Johannes D. Meeldijk and Sara Bals and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84921731109&doi=10.1021%2fcm504340h&partnerID=40&md5=c6a111289f1683b897c4549634cc0919},
doi = {10.1021/cm504340h},
year = {2015},
date = {2015-01-01},
journal = {Chemistry of Materials},
volume = {27},
number = {2},
pages = {621 – 628},
note = {Cited by: 122; All Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Akkerman, Quinten A.; Ke, Xiaoxing; Huis, Marijn A. Van; Bals, Sara; Donega, Celso De Mello
Solution-processable ultrathin size- and shape-controlled colloidal Cu2- xS nanosheets Journal Article
In: Chemistry of Materials, vol. 27, no. 1, pp. 283 – 291, 2015, (Cited by: 71; All Open Access, Green Open Access).
@article{VanDerStam2015283b,
title = {Solution-processable ultrathin size- and shape-controlled colloidal Cu2- xS nanosheets},
author = { Ward Van Der Stam and Quinten A. Akkerman and Xiaoxing Ke and Marijn A. Van Huis and Sara Bals and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84921348715&doi=10.1021%2fcm503929q&partnerID=40&md5=22b8b3fe5d0c2bb8f9916861ffbbc219},
doi = {10.1021/cm503929q},
year = {2015},
date = {2015-01-01},
journal = {Chemistry of Materials},
volume = {27},
number = {1},
pages = {283 – 291},
note = {Cited by: 71; All Open Access, Green Open Access},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, W Van Der; Gantapara, A P; Akkerman, Q A; Soligno, G; Meeldijk, J D; Roij, R Van; Dijkstra, M; Donega, C De Mello
Self-assembly of colloidal hexagonal bipyramid- and bifrustum-shaped ZnS nanocrystals into two-dimensional superstructures Journal Article
In: Nano Letters, vol. 14, no. 2, pp. 1032-1037, 2014, (cited By 62).
@article{VanDerStam20141032,
title = {Self-assembly of colloidal hexagonal bipyramid- and bifrustum-shaped ZnS nanocrystals into two-dimensional superstructures},
author = {W Van Der Stam and A P Gantapara and Q A Akkerman and G Soligno and J D Meeldijk and R Van Roij and M Dijkstra and C De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84894141500&doi=10.1021%2fnl4046069&partnerID=40&md5=77c3dc3df690fa29437a898fa7d800ee},
doi = {10.1021/nl4046069},
year = {2014},
date = {2014-01-01},
journal = {Nano Letters},
volume = {14},
number = {2},
pages = {1032-1037},
note = {cited By 62},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Stam, Ward Van Der; Gantapara, Anjan P.; Akkerman, Quinten A.; Soligno, Giuseppe; Meeldijk, Johannes D.; Roij, René Van; Dijkstra, Marjolein; Donega, Celso De Mello
Self-assembly of colloidal hexagonal bipyramid- and bifrustum-shaped ZnS nanocrystals into two-dimensional superstructures Journal Article
In: Nano Letters, vol. 14, no. 2, pp. 1032 – 1037, 2014, (Cited by: 76).
@article{VanDerStam20141032b,
title = {Self-assembly of colloidal hexagonal bipyramid- and bifrustum-shaped ZnS nanocrystals into two-dimensional superstructures},
author = { Ward Van Der Stam and Anjan P. Gantapara and Quinten A. Akkerman and Giuseppe Soligno and Johannes D. Meeldijk and René Van Roij and Marjolein Dijkstra and Celso De Mello Donega},
url = {https://www.scopus.com/inward/record.uri?eid=2-s2.0-84894141500&doi=10.1021%2fnl4046069&partnerID=40&md5=77c3dc3df690fa29437a898fa7d800ee},
doi = {10.1021/nl4046069},
year = {2014},
date = {2014-01-01},
journal = {Nano Letters},
volume = {14},
number = {2},
pages = {1032 – 1037},
note = {Cited by: 76},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Other publications
See earlier publications on Scopus.


